TLIF: A Guide to One of Modern Spine Surgery's Most Established Techniques

Transforaminal Lumbar Interbody Fusion
A guide to one of modern spine surgery's most established techniques
From open surgery to endoscopy • 1998 → today
1. What is a TLIF?
Transforaminal Lumbar Interbody Fusion — TLIF — is a surgical technique used to permanently join two adjacent lumbar vertebrae. The goal is to eliminate painful motion at a degenerated or unstable disc level while preserving, and often improving, the alignment of the spine.
The procedure is built on three combined steps. First, the surgeon approaches the spine from behind (a posterior approach) and removes one facet joint on a single side. That single-sided removal opens a narrow corridor through the intervertebral foramen — the natural opening through which the spinal nerve exits — and gives access to the disc space without crossing the midline of the spinal canal.
Second, the damaged intervertebral disc is removed and replaced with an interbody cage filled with bone graft. The cage restores disc height, decompresses the exiting nerve, and provides a scaffold for bone to grow from one vertebra to the next.
Third, pedicle screws are placed on both sides and connected by rods. This construct immobilizes the segment while bone fusion takes place over the following months — typically six to twelve.
Why “transforaminal”?
Earlier posterior fusion techniques (PLIF) accessed the disc by retracting the dural sac — the nerve-containing membrane — from one side to the other. TLIF was designed specifically to avoid that maneuver. By entering through the foramen on one side only, the surgeon reaches the disc without pulling on the nerves of the cauda equina. This single design choice is the reason TLIF was adopted so rapidly worldwide.

2. A short history: how surgeons arrived at the TLIF
TLIF did not appear in a vacuum. It is the result of nearly a century of trial, error, and stepwise improvement in lumbar spine surgery.
2.1 1934 — Identifying the problem
In August 1934, two Boston physicians, William J. Mixter and Joseph S. Barr, published a landmark paper in the New England Journal of Medicine. They demonstrated that lumbar disc herniation — not bony abnormalities alone — was a major cause of sciatica and lumbar instability. Their work shifted the entire field's attention toward the intervertebral disc as a surgical target.
2.2 1944–1953 — The birth of posterior interbody fusion (PLIF)

In 1944, Briggs and Milligan described “chip fusion”: after exposing the spine posteriorly, they removed bony chips, partially excised the lamina and facets, removed the disc, and replaced it with a round bone peg. The Hawaiian neurosurgeon Ralph Cloward refined and popularized the technique in 1953 under the name Posterior Lumbar Interbody Fusion (PLIF). PLIF worked, but it required bilateral retraction of the dural sac, exposing nerves to traction injury, and it was technically demanding.
2.3 1970s–1980s — Pedicle screws and Kambin's triangle
Two parallel innovations transformed lumbar surgery in this period. Raymond Roy-Camille, in early-1970s Paris, introduced pedicle screw fixation — a three-column construct that gave surgeons unprecedented stability while bone fused. Around the same time, Philadelphia surgeon Parviz Kambin described a working anatomical “triangle” within the foramen: a small zone bounded by the exiting root, the traversing root, and the superior endplate, free of major neural structures and theoretically safe for instrument passage. Kambin's triangle would later become the gateway for endoscopic fusion.
2.4 1982 → 1998 — Harms and Rolinger invent the TLIF
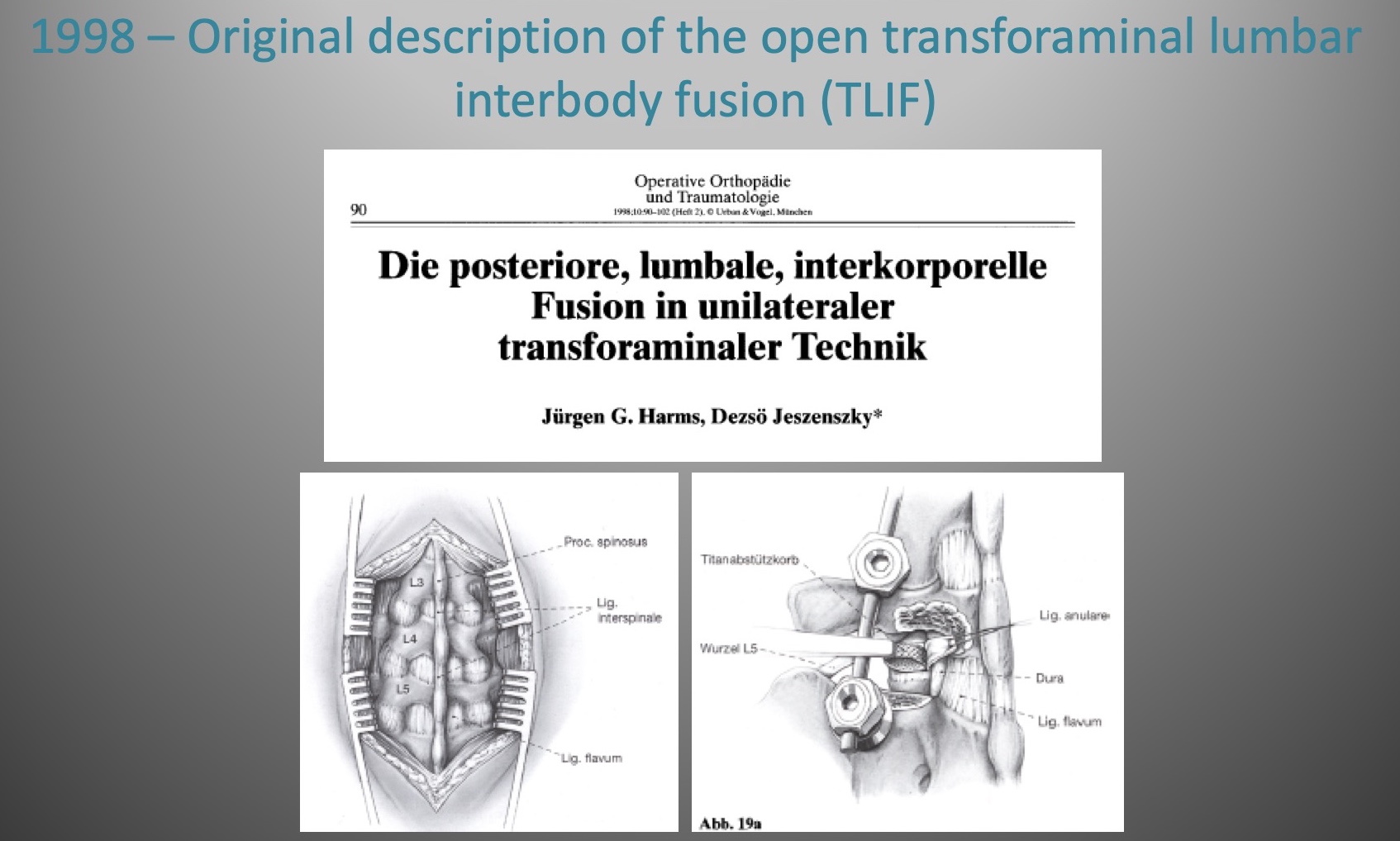
In 1982, two German surgeons — Jürgen Harms and H. Rolinger — proposed a fundamentally new posterior approach. Instead of crossing the spinal canal bilaterally as in PLIF, they accessed the disc through the foramen on a single side after removing one facet. They packed a titanium mesh cage with bone graft and placed it in the anterior disc space. In 1998, Harms and Jeszenszky published the refined, complete-facetectomy version that became the modern open TLIF. The procedure was immediately attractive: less nerve retraction, less dural manipulation, full three-column support from a single incision.
2.5 2002 — The minimally invasive revolution
By the late 1990s spine surgeons had begun to question the cost of open exposures: large incisions, paraspinal muscle stripping, prolonged back pain. In 2002, Kevin Foley and Michael Lefkowitz published the Minimally Invasive TLIF (MIS-TLIF), performed through two small paramedian incisions and a tubular muscle-splitting retractor. The surgical principle remained Harms's; the surgical footprint shrank dramatically.
2.6 2010s–today — The endoscopic era
The success of endoscopic discectomy in the 2010s inspired surgeons to attempt full interbody fusion through endoscopic portals. Endoscopic TLIF (Endo-TLIF), intra-foraminal LIF (iLIF) through Kambin's triangle, and unilateral biportal endoscopic TLIF (UBE-TLIF) emerged in rapid succession. The latest generation of techniques can be performed under conscious sedation in selected cases — a milestone unimaginable in 1998.
3. The TLIF family: three techniques, one principle
Today, three main variants of TLIF coexist. All three share the same surgical philosophy — a unilateral transforaminal route to the disc — but differ markedly in incision size, visualization tools, and the patient experience around surgery.
3.1 Open TLIF
The classical technique. Performed through a single midline incision typically 8–12 cm long. The paraspinal muscles are stripped from the spine to expose the lamina and facets bilaterally. The surgeon works under direct vision, with excellent anatomical orientation. Open TLIF remains the reference for complex deformity, revision surgery, and multi-level constructs.
Strengths: wide direct visualization, easier multi-level work, well-suited to deformity correction and posterior osteotomies.
Limitations: extensive muscle dissection, higher blood loss, more postoperative pain, longer hospital stay.
3.2 Minimally invasive TLIF (MIS-TLIF)
Introduced by Foley and Lefkowitz in 2002. Two paramedian incisions of about 2.5–3 cm each. A sequence of progressively larger tubular dilators is passed through the natural cleavage plane of the paraspinal muscles (the Wiltse plane) until a working tubular retractor is docked on the facet joint. Decompression, facetectomy, discectomy, and cage placement are performed through the tube under microscope or loupe magnification with fluoroscopic guidance. Percutaneous pedicle screws are then placed on both sides.
Strengths: preservation of paraspinal muscles, reduced blood loss, shorter hospital stay, faster early recovery, reduced postoperative pain.
Limitations: narrow visual field, steep learning curve, increased intraoperative fluoroscopy and therefore radiation exposure, technical difficulty with lordosis restoration at L5–S1.
3.3 Endoscopic TLIF (Endo-TLIF, iLIF, UBE-TLIF)
The newest member of the family. Surgery is performed under continuous saline irrigation through one or two small portals (8–10 mm) using a rigid endoscope. Variants differ by approach geometry: uniportal endoscopic TLIF and intra-foraminal LIF (iLIF) use a single working channel through Kambin's triangle while sparing the inferior articular process; biportal endoscopic TLIF (UBE-TLIF) uses two separate portals — one for the endoscope, one for instruments — and allows wider bone work. Some centers now perform endoscopic TLIF under spinal, epidural, or even conscious sedation, allowing real-time neurological monitoring by direct patient questioning.
Strengths: smallest tissue footprint, lowest blood loss, shortest hospital stay (often outpatient or 24–48 h), reduced or eliminated general anesthesia in selected cases, direct endoscopic visualization of the exiting nerve root.
Limitations: very steep learning curve; restricted access to the disc space limits cage size; long-term comparative outcome data still maturing.
Side-by-side comparison
| Parameter | Open TLIF | MIS-TLIF | Endoscopic TLIF |
|---|---|---|---|
| Parameter | Open TLIF | MIS-TLIF | Endoscopic TLIF |
| Incision | Single midline, 8–12 cm | Two paramedian, ~3 cm | One or two portals, 8–10 mm |
| Muscle damage | Extensive stripping | Muscle-splitting | Minimal |
| Typical blood loss | 250–500 mL | 100–250 mL | < 100 mL |
| Hospital stay | 4–7 days | 2–4 days | 1–2 days (sometimes outpatient) |
| Visualization | Direct, wide | Tubular + microscope | Endoscope, saline-irrigated |
| Anesthesia | General | General | General, spinal, or sedation |
| Fluoroscopy | Low | Higher | Higher |
| Learning curve | Moderate | Steep | Very steep |
| Multi-level / deformity | Best suited | Selected cases | Generally single-level |
Table 1. Practical comparison of the three contemporary TLIF variants. Figures are typical orders of magnitude and depend on operator experience, patient anatomy, and number of levels treated.
4. Inside the operation: the TLIF step by step
Beyond the choice between open, minimally invasive, or endoscopic technique, every TLIF follows the same essential surgical sequence. The illustrations below pair the classical anatomical drawings with intraoperative photographs of the corresponding step in a real MIS-TLIF case.
Step 1 — Pedicle screw placement and facetectomy
After patient positioning and surgical approach, the surgeon places pedicle screws bilaterally under fluoroscopic guidance. On the symptomatic side, a complete facetectomy is performed: the inferior articular process of the upper vertebra is removed, exposing the underlying superior articular process of the lower vertebra. The harvested bone is preserved as autograft for later use inside the cage.

Step 2 — Superior facet resection
The superior articular process of the lower vertebra is then resected. This is the key step that distinguishes TLIF from earlier posterior techniques: by removing this single bony structure, the surgeon opens the foramen widely on one side and gains direct access to the lateral disc space without ever entering the spinal canal or retracting the dura.

Step 3 — Disc exposure
Through the now-open foramen, the exiting nerve root is identified and gently protected. The disc — appearing as a glistening fibrous structure between the two vertebral bodies — comes into view in the working window. The annulus fibrosus is then incised to enter the disc space.

Step 4 — Endplate preparation
Endplate preparation is the most underappreciated step of a successful fusion. Using curettes, rasps, and specialized shavers, the surgeon removes the entire nucleus pulposus and meticulously decorticates both the upper and lower vertebral endplates. The aim is to expose bleeding cancellous bone — the biological substrate without which no fusion can occur — while preserving the cortical rim that will support the cage and prevent subsidence.

Step 5 — Cage placement
The prepared disc space is then packed anteriorly with morcellized autograft bone (typically from the resected facets). The interbody cage — filled with additional bone graft — is inserted through the foraminal corridor and rotated into position in the anterior third of the disc space. Final position is confirmed by fluoroscopy. The cage restores disc height, distracts the foramen to decompress the exiting nerve, and provides immediate mechanical support.

Step 6 — Final construct
Two rods are then connected to the pedicle screws, with mild compression to maximize cage–endplate contact and to restore segmental lordosis. After meticulous hemostasis and closure, the construct provides immediate stability while the bone fusion process — typically complete between six and twelve months — takes place inside and around the cage.
5. When is TLIF indicated?
TLIF is offered when a degenerated or unstable lumbar disc produces persistent pain or neurological symptoms that have not responded to several months — usually at least three to six — of well-conducted conservative care: physiotherapy, targeted exercise, oral analgesics, and, where appropriate, image-guided injections. Surgery is never the first-line answer; it is the answer when non-surgical treatment has been given a real chance and has failed.
Within that frame, TLIF addresses a broad spectrum of degenerative and structural pathologies of the lumbar spine.
5.1 Degenerative spondylolisthesis
An age-related forward slippage of one vertebra over the one below it, most often at L4–L5, caused by progressive arthritis of the facet joints and disc degeneration. When the slippage produces mechanical low back pain, neurogenic claudication, or radicular leg pain unresponsive to conservative care, decompression combined with TLIF restores foraminal height, addresses the disc, and stabilizes the segment in a single procedure.
5.2 Isthmic spondylolisthesis
Caused by a defect in the pars interarticularis (the bony bridge between superior and inferior facets), most often at L5–S1. The vertebra slides forward through the defect. TLIF allows restoration of disc height, correction of the slippage, and circumferential fusion through a single posterior approach — often particularly relevant in younger adults.
5.3 Degenerative disc disease with mechanical instability
A symptomatic, painful disc with dynamic instability on flexion-extension imaging is one of the historical indications for TLIF. The fused interbody segment eliminates the painful micromotion.
5.4 Lumbar spinal stenosis with instability
When central, lateral recess, or foraminal narrowing produces neurogenic claudication or radicular pain and is associated with segmental instability — or when decompression alone would require destabilizing bony resection — TLIF provides both decompression and fusion.
5.5 Recurrent disc herniation
A second herniation at a previously operated level often signals more advanced disc failure. When microdiscectomy alone is unlikely to provide durable relief, TLIF replaces the disc entirely and stabilizes the level.
5.6 Failed back surgery syndrome
Persistent or recurrent pain after previous lumbar surgery, when imaging shows a structurally treatable cause (pseudarthrosis, residual instability, adjacent-segment degeneration), is a recognized indication. Revision is technically more demanding and usually performed through an open approach.
5.7 Selected fractures, tumors, and infections
Certain traumatic burst fractures with kyphotic collapse, selected localized tumors, and stabilized spinal infections can benefit from TLIF combined with adapted decompression. These are specialist indications and outside the scope of a routine TLIF discussion.
Contra-indications and cautions
Severe osteoporosis (compromises screw fixation and increases subsidence), active untreated infection at the surgical site, certain severe deformities requiring anterior or lateral approaches, and unstable major medical comorbidities. Each case is assessed individually — TLIF is one tool in a wider toolbox that also includes ALIF, OLIF, LLIF, and posterolateral fusion.
6. What does the most recent evidence say?
The literature on TLIF is now mature: more than four decades of accumulated experience, multiple randomized controlled trials, and several major systematic reviews. The signal that emerges from the most recent meta-analyses is consistent.
6.1 TLIF works — and the technique is converging
Across degenerative indications, TLIF produces clinically meaningful improvements in disability (Oswestry Disability Index) and pain (Visual Analog Scale), with fusion rates typically above 90% at one year. A 2022 systematic review and meta-analysis by Wasinpongwanich and colleagues pooled 15 randomized controlled trials and 915 patients comparing TLIF to other techniques (PLF, PLIF, ALIF, XLIF). It found broadly equivalent clinical outcomes across approaches, with TLIF showing a slightly longer operative time but comparable fusion rates at two years and similar complication profiles.
6.2 Endoscopic TLIF: a serious contender
The 2022 systematic review and meta-analysis by Sousa et al. (Scientific Reports), pooling 42 studies and over 2,200 patients comparing endoscopic intra-foraminal LIF (iLIF) to MIS-TLIF, delivered four key findings:
Significantly lower intraoperative blood loss with iLIF (mean difference ≈ 110 mL favoring iLIF).
Significantly shorter hospital stay (mean difference ≈ 2.4 days favoring iLIF).
Equivalent clinical improvement in VAS back, VAS leg, and ODI scores — both techniques produced clinically meaningful gains.
No significant difference in fusion rates or overall complication rates.
The authors concluded that iLIF, when performed by experienced operators, provides outcomes at least equivalent to MIS-TLIF with a measurable reduction in surgical morbidity. Their data also support performing the procedure under regional anesthesia or sedation in selected patients, with intraoperative neurological monitoring made possible by direct patient questioning.
6.3 MIS-TLIF and long-term outcomes: the Denisov 2026 meta-analysis
The most important recent contribution to the TLIF literature — and the one that anchors the registry research described in Section 7 — is the meta-analysis published in January 2026 in Spine Open by Anton Denisov and colleagues. The question they set out to answer was not whether MIS-TLIF works in the perioperative period (settled long ago) but whether the perioperative advantages of MIS translate into better long-term outcomes — specifically, adjacent segment disease (ASD) and reoperation.
ASD is the late “price” of any spinal fusion. By eliminating motion at one level, fusion transfers mechanical load to the discs and facets immediately above and below. Some of those adjacent segments degenerate symptomatically over the following years and may require further surgery. Whether MIS-TLIF, by sparing the paraspinal muscles and posterior tension band, reduces that long-term burden has been debated for two decades. Earlier meta-analyses had been inconclusive, partly because they pooled studies with short follow-up.
Methods and scope
Denisov and colleagues registered their protocol with PROSPERO and followed PRISMA guidelines. They searched six databases (PubMed, Cochrane, Scopus, Embase, Web of Science, Google Scholar) for studies published between January 2000 and June 2024 comparing MIS-TLIF to open TLIF or PLIF in degenerative lumbar disease, with a minimum follow-up of 24 months. Random-effects meta-analyses were performed with multivariate meta-regression for patient-level predictors. Risk of bias was assessed with ROBINS-I and certainty of evidence with GRADE. Fourteen studies met all inclusion criteria, encompassing 1,960 patients (909 MIS-TLIF, 1,051 open TLIF/PLIF). Follow-up ranged from 24 to 120 months.
Key results
| Outcome | Odds Ratio (95% CI) | p-value | Heterogeneity (I²) |
|---|---|---|---|
| Outcome | Odds Ratio (95% CI) | p-value | Heterogeneity (I²) |
| Adjacent segment disease (ASD) | 0.51 (0.37–0.69) | < 0.001 | 0 % |
| Reoperation (any cause) | 0.53 (0.31–0.90) | 0.018 | 12.6 % |
| Pseudarthrosis (non-union) | 0.96 (0.38–2.48) | 0.94 | 0 % |
| ODI improvement (disability) | Comparable between groups | ns | — |
| VAS back pain improvement | Greater reduction with MIS in 2/5 studies | 0.02–0.05 | — |
Table 2. Principal findings of the Denisov et al. 2026 meta-analysis (14 studies, 1,960 patients, ≥ 24 months follow-up). Odds ratio < 1 favours MIS-TLIF. Source: Denisov A, Zaborovskii N, Elshoeibi AM, Tornero E, Hamouda WO, Alzahrani AJ, Aly MM. Spine Open 2026;2:e0043.
The headline numbers are unambiguous. MIS-TLIF was associated with a 49 % relative reduction in the odds of adjacent segment disease and a 47 % reduction in the odds of reoperation compared with open TLIF/PLIF, with essentially no heterogeneity between studies (I² = 0 % for ASD). The protective effect on ASD held consistently in both follow-up subgroups (24 months and beyond 24 months) and survived a sensitivity analysis restricted to studies at low or moderate risk of bias (OR 0.52, 95 % CI 0.36–0.77). At the same time, fusion success was identical between the two approaches — the perioperative advantage of MIS does not come at the cost of weaker bony healing.
Patient-level predictors of ASD
Beyond the comparison between approaches, the meta-regression identified two patient-level factors that drive ASD risk independently of how the surgery was performed:
Multilevel fusion (≥ 2 levels vs single level): OR 3.32 (95 % CI 1.54–7.11), p = 0.002. Each additional fused level substantially raises the long-term load transferred to the adjacent segments. This finding reaffirms a long-standing biomechanical principle.
Older age — protective: OR 0.85 per year (95 % CI 0.78–0.93), p < 0.001. This is at first counter-intuitive (older patients generally have more degeneration), but it likely reflects shorter constructs, lower postoperative activity levels, and reduced motion-related stress in older patients.
BMI showed a non-significant trend toward lower risk (p = 0.068), smoking a non-significant trend toward higher risk (p = 0.071), and follow-up duration was not an independent predictor.
Why this matters
Denisov 2026 shifts a previously contested clinical question into the settled column: for degenerative lumbar disease, MIS-TLIF reduces the long-term complications that matter most to patients — the need for another operation — by roughly half, with the same fusion success and the same functional gain. The number of levels fused, and the patient's age, are now confirmed independent drivers of long-term risk. These two predictors form the backbone of the patient-specific risk modeling described in Section 7.
6.4 Cage materials: PEEK, titanium, 3D-printed titanium
The interbody cage is no longer a passive spacer. Three families dominate today's market:
Polyetheretherketone (PEEK): radiolucent, with a Young's modulus close to cortical bone. Subsidence rates are moderate (≈ 15–20 %), but PEEK is biologically inert — bone does not adhere to its surface, and small zones of fibrous tissue can form at the bone–cage interface.
Solid titanium: excellent osseointegration and high fusion rates, but a stiffness much higher than bone, leading to historically elevated subsidence rates (up to 35 % in some series).
3D-printed porous titanium: the newest entrant. Additive manufacturing produces a lattice that mimics trabecular bone, lowering the apparent stiffness while preserving titanium's biological advantages. A 2022 comparative study (Kim et al., Neurospine) and a 2024 network meta-analysis suggest higher early fusion grades versus PEEK at one year, with no excess in subsidence.
Expandable cages, which can be deployed in a low profile and then expanded inside the disc, are increasingly used to restore segmental lordosis through the narrow MIS or endoscopic working channel. The radiographic gain is real; long-term subsidence data are still maturing.
6.5 Where the field is going
Three trends are reshaping TLIF in real time. Robotic-assisted pedicle screw placement is improving accuracy and reducing radiation exposure. Intraoperative navigation — including augmented-reality head-mounted displays — is starting to compensate for the visualization deficit of minimally invasive corridors. And cage engineering is moving toward patient-specific, bioactive, 3D-printed devices designed to favor osseointegration.
The combined direction of travel is clear: smaller incisions, less general anesthesia, more accurate instrumentation, and smarter implants — without sacrificing the surgical principle Harms defined in 1998.
7. TLIF-BAYES: turning meta-analytic evidence into patient-specific prediction
The Denisov 2026 meta-analysis answered a population-level question: across thousands of patients, MIS-TLIF roughly halves the odds of adjacent segment disease and reoperation compared with open TLIF. But meta-analyses, by construction, produce a single average answer for an average patient. They cannot tell a 58-year-old non-smoker with a BMI of 29 and 14° of pelvic–lumbar imbalance what her individual risk would be if she chose one approach over another.
TLIF-BAYES is the next logical step. Led by the same investigator who anchored the meta-analysis, Dr. Anton Denisov, in collaboration with SpineDAO and the SpineBase international spine surgery registry, the study moves from aggregate evidence to individualized prediction. The methodological link between the two studies is unusually clean: the odds ratio from the meta-analysis becomes the informative prior for the registry's Bayesian model.
7.1 Study design at a glance
| Principal Investigator | Anton Denisov, MD, M.Biostat |
| Sponsor / platform | SpineDAO · SpineBase International Spine Surgery Registry |
| Data platform | spinebase.app |
| Study type | Multicenter registry-based observational cohort, retrospective + prospective |
| Surgical arms | Open TLIF · MIS-TLIF · Endoscopic TLIF |
| Statistical platform | R (RStudio) + Stan (brms) for Bayesian hierarchical modeling |
| Protocol version | v1.1 — March 2026 (supersedes v1.0, January 2026) |
| Current enrollment | 700 TLIF patients across contributing centres |
| Target | 1000+ patients for primary outcome analysis (July 2026); 2,000+ for cause-stratified analyses |
Table 3. TLIF-BAYES study at a glance. Source: TLIF-BAYES Protocol v1.1, SpineDAO / SpineBase, March 2026.
7.2 What the study is asking
The protocol formulates the question in three layers. The primary objective is to estimate the probability of revision surgery following TLIF using Bayesian hierarchical modeling, with the cause of each revision classified by a structured nine-code taxonomy. The secondary objectives extend the analysis to a direct comparison of open, minimally invasive, and endoscopic arms; identification of patient-level predictors of postoperative complications; longitudinal patient-reported outcomes (ODI, VAS back, VAS leg) over two years; the development of a patient-specific clinical risk prediction tool; and a mechanistic analysis of whether sagittal correction (the change in pelvic–lumbar mismatch) modifies long-term risk.
7.3 Why revision surgery, not symptomatic ASD
Protocol v1.1 made a deliberate change from v1.0: the primary outcome is no longer “symptomatic adjacent segment disease” but “revision surgery, classified by structured cause taxonomy.” The reasoning is practical. Symptomatic ASD is a composite construct (radiographic degeneration plus clinical symptoms) that is reliably defined prospectively but very hard to ascertain retrospectively across heterogeneous centres. Revision surgery, by contrast, is always documented, always binary, and always has a cause recorded in the operative report. ASD-related revision is preserved as a pre-specified subgroup analysis — directly corresponding to the endpoint of the Denisov meta-analysis.
Each revision is coded into one of nine categories: adjacent segment disease (ASD), pseudarthrosis, infection, screw malposition, dural tear / CSF leak, cage migration, hematoma, new neurological deficit, or other (with mandatory free-text detail). This taxonomy lets the model characterize the full reoperation landscape rather than collapsing every late event into a single composite.
7.4 The Bayesian link to Denisov 2026
From meta-analysis to prior
In a Bayesian analysis, a “prior” is the belief about an effect before new data are considered. In TLIF-BAYES, the prior for the effect of MIS versus open TLIF is set to Normal(log 0.51, 0.20) — a distribution centred exactly on the odds ratio of 0.51 published by Denisov et al. in 2026, with a width reflecting the precision of that estimate. The new registry data then update this prior into a posterior distribution. This is the cleanest possible methodological lineage: the same investigator's published evidence is used, transparently and quantitatively, as the starting point for the next layer of analysis. The endoscopic arm, for which no published patient-level comparator exists, is given a weakly informative prior centred on the null.
7.5 Patient-level predictors built into the model
Beyond the surgical approach, the model accounts for the patient-level factors known to drive long-term TLIF outcomes — several of which were confirmed by the Denisov meta-analysis itself:
Demographics: age (confirmed predictor in Denisov 2026), BMI, smoking status.
Construct: number of fused levels (confirmed strong predictor in Denisov 2026, OR 3.32 for multilevel).
Sagittal alignment: preoperative pelvic incidence – lumbar lordosis (PI–LL) mismatch, with postoperative PI–LL added as a sensitivity covariate to assess whether surgical correction modifies risk.
Surgical quality: facet joint violation graded on the Babu ordinal 0–3 CT-based scale.
Exploratory predictors (already captured in the 245-patient dataset, novel at patient level in the TLIF literature): Pfirrmann grade of the adjacent disc, Modic changes at the adjacent endplate, Schizas grade of foraminal stenosis. No published patient-level TLIF outcomes study has previously included these as predictors of adjacent segment reoperation.
Hierarchical structure: surgeon and centre are modeled as random effects, allowing the analysis to separate the signal of approach choice from the noise of who performed the surgery and where.
7.6 Fusion assessment: a two-tier framework
Pseudarthrosis (failed bony fusion) is assessed in two tiers. Tier 1 is plain radiograph assessment, available for the full cohort: a simplified binary read — fused / not fused / indeterminate — derived from the Bridwell grading system. Tier 2 is CT-confirmed pseudarthrosis using the Brantigan–Steffee–Fraser (BSF) classification, available for centres with routine post-operative CT. Fusion assessment modality and date are recorded for every patient and enter the model as covariates, so that heterogeneity in diagnostic sensitivity across centres becomes an explicit part of the analysis rather than a hidden source of bias.
7.7 The clinical deliverable: patient-specific risk prediction
The endpoint of TLIF-BAYES is not a single p-value. It is a deployed clinical tool. Posterior distributions from the hierarchical model will be used to generate individualized risk predictions, implemented as a web application on spinebase.app. A surgeon planning a TLIF will be able to enter a patient's profile and obtain a counterfactual estimate of revision risk under each of the three surgical approaches.
Example output (illustrative, from the protocol)
Patient: 58 years old, BMI 29, non-smoker, single-level TLIF, PI–LL mismatch 14°, no facet violation. • Overall revision risk after open TLIF: 12 % (95 % credible interval 7–19 %) • Revision for ASD specifically: 6 % (95 % CI 3–11 %) • Pseudarthrosis risk: 4 % (95 % CI 2–8 %) • Counterfactual: same patient, MIS approach → overall revision risk 7 % (95 % CI 3–13 %).
7.8 Timeline and deliverables
| Milestone | Date |
|---|---|
| Milestone | Date |
| Protocol v1.0 | January 2026 |
| Denisov meta-analysis published (Spine Open) — source of informative prior | 23 January 2026 |
| Protocol v1.1 (current), incorporating four amendments | March 2026 |
| First Bayesian model run on available data (exploratory) | April 2026 |
| Target 600 patients for primary outcome analysis | April 2026 |
| Presentation at the NSpine Southern European Summit, Bastia | May 2026 |
| Target 1,000+ patients for reliable cause-stratified analysis | Rolling thereafter |
Table 4. TLIF-BAYES timeline and key deliverables. Source: TLIF-BAYES Protocol v1.1, Section 13.
7.9 Ethics, data, and dissemination
The study uses de-identified registry data collected via the SpineBase platform, with HIPAA Safe Harbor and GDPR-compliant de-identification. Ethics approval follows the institutional requirements of each participating centre; retrospective de-identified data are typically exempt from full IRB review, and SpineDAO provides template exemption language. Results will be disseminated through peer-reviewed publications, international spine conferences — including the NSpine Southern European Summit in Bastia, May 2026 — and a clinical risk prediction calculator integrated directly into spinebase.app.
How to participate
Centres performing open, minimally invasive, or endoscopic TLIF for degenerative lumbar disease can contribute to TLIF-BAYES through the SpineBase platform at spinebase.app. Contribution guides for new centres are being distributed during March–April 2026 ahead of the first Bayesian model run.
— End of document —
Key references
Harms JG, Jeszenszky D. Die posteriore, lumbale, interkorporelle Fusion in unilateraler transforaminaler Technik. Oper Orthop Traumatol 1998;10:90–102.
Foley KT, Lefkowitz MA. Advances in minimally invasive spine surgery. Clin Neurosurg 2002;49:499–517.
de Kunder SL, Rijkers K, Caelers IJMH, et al. Lumbar interbody fusion: a historical overview and a future perspective. Spine 2018;43:1161–8.
Sousa JM, Ribeiro H, Silva JL, Nogueira P, Consciência JG. Clinical outcomes, complications and fusion rates in endoscopic assisted intraforaminal lumbar interbody fusion (iLIF) versus minimally invasive transforaminal lumbar interbody fusion (MI-TLIF): systematic review and meta-analysis. Sci Rep 2022;12:2101.
Wasinpongwanich K, Nopsopon T, Pongpirul K. Surgical treatments for lumbar spine diseases (TLIF vs. other surgical techniques): a systematic review and meta-analysis. Front Surg 2022;9:829469.
Hammad A, Wirries A, Ardeshiri A, et al. Open versus minimally invasive TLIF: literature review and meta-analysis. J Orthop Surg Res 2019;14:229.
Prabhu MC, Jacob KC, Patel MR, Pawlowski H, Vanjani NN, Singh K. History and evolution of the minimally invasive transforaminal lumbar interbody fusion. Neurospine 2022;19(3):479–91.
Verma R, Virk S, Qureshi S. Interbody fusions in the lumbar spine: a review. HSS J 2020;16:162–7.
Virk S, Qureshi S, Sandhu H. History of spinal fusion: where we came from and where we are going. HSS J 2020;16:137–42.
Kim DY, Kwon OH, Park JY. Comparison between 3-dimensional-printed titanium and polyetheretherketone cages: 1-year outcome after minimally invasive transforaminal interbody fusion. Neurospine 2022;19(3):524–32.
Denisov A, Zaborovskii N, Elshoeibi AM, Tornero E, Hamouda WO, Alzahrani AJ, Aly MM. Does minimally invasive TLIF reduce adjacent segment disease and reoperation rates compared with open TLIF? A meta-analysis. Spine Open 2026;2:e0043.
Denisov A (PI). TLIF-BAYES — Bayesian hierarchical modeling and individualized prediction of outcomes after open, minimally invasive, and endoscopic transforaminal lumbar interbody fusion. Study Protocol v1.1, SpineDAO / SpineBase, March 2026.













%20Medium.png)


.png)
